The choice candy for the holiday season makes for an awesome science experiment! Our dissolving candy cane experiments are an easy and frugal Christmas science experiment.

#1 Candy Cane Experiment: Temperature of Water
Variation: I was trying to decide if we should use the candy canes or the peppermints, so my son suggested we do both. Then I suggested we weigh the candy cane and the peppermint to see if they were the same weight.
We discovered that both candies are the same weight but differ in shape. We used a kitchen scale and had the opportunity to discuss the numbers and measurements between ounces and grams.
💡 How will the shapes of the peppermint and the candy cane affect the results? Which will dissolve faster? Make a prediction, set up an experiment, and test your theory. You can read more about the scientific method for kids here.
You Will Need:
- Small Candy Canes
- Small peppermints {Optional}
- Water
- Cups
- Stopwatch/Timer and/or Kitchen Scale
- Printable Science Worksheet {scroll down}
Experiment #1 Set Up
STEP 1. Fill your cups with the same amount of water but at different temperatures. Make sure to label what you have in each cup.
We chose room temperature water, boiled water from the kettle, and freezer cold water.
WARNING: Younger kids will require adult assistance for handling very hot water!
STEP 2. Add one candy cane or peppermint to each cup. Make sure you add the same type of candy cane to each cup.
Optional: Makeup two cups of each type of liquid if you want to compare candy canes and round peppermints.
STEP 3. Set the timer to record how long each peppermint or candy cane takes to dissolve.
STEP 4. Observe what happens.
Please download our candy cane science worksheet below to record your results.

Candy Cane Experiment Worksheet
Add this dissolving candy cane experiment page to a science journal to extend the activity for older kids!
#2 Candy Cane Experiment: Different Liquids
This candy cane experiment explores how fast the candy cane dissolves in different solutions that you can easily make up for yourself, salt water and sugar water.
How will the type of liquid affect the results? Which will dissolve faster?
You Will Need:
- 6 cups of water
- ½ cup sugar, divided
- ½ cup salt, divided
- 6 candy canes
#2 Candy Cane Experiment Set Up
STEP 1. To make your solutions… Add 1 cup of water to three different cups. Then add ¼ cup sugar to one of the cups, stirring until it is dissolved. Add ¼ cup salt to the second cup, stirring until dissolved. The third cup is the control.

STEP 2. Heat another 3 cups of water until hot. Place 1 cup of hot water into another three cups. Into one of these cups, add ¼ cup sugar, stirring until it is dissolved. Into the second cup with hot water, add ¼ cup salt, stirring until dissolved. The third cup is the control.
STEP 3. Place one unwrapped candy cane into each cup of water. Set a timer for 2 minutes.

When the timer goes off, check the candy canes and make note of which have changed. Continue checking the candy canes every 2 to 5 minutes, making note of the changes.
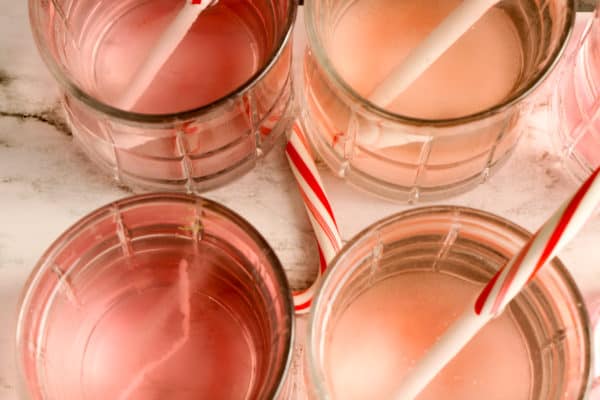
Discuss which liquids caused the candy canes to dissolve faster/slower and why.
If desired, repeat the experiment using different room-temperature liquids such as vinegar, liquid dish soap, oil, soda pop, etc.

Why Do Candy Canes Dissolve?
Water vs Oil
Candy canes are made up of sugar molecules! Sugar dissolves in water because energy is given off when the sucrose molecules (which make up sugar) form bonds with the water molecules. The sugar molecules attract water molecules and if powerful enough of an attraction, will separate and dissolve!
What about oil? The sugar in candy canes is soluble in water but not in oil. You would have noticed that when you place a candy cane in oil, it won’t dissolve in the same way it would in water.
The solubility of a substance depends on the nature of the solvent (the substance in which the solute dissolves), and sugar, the main component of candy canes, is hydrophilic (water-attracting) but hydrophobic (oil-repelling).
The Effect of Temperature
The solubility of substances, including candy canes, generally increases with temperature. In hot water, molecules move more rapidly than in cold water because they have higher kinetic energy. This increased movement and energy lead to more collisions between water molecules and sugar molecules on the surface of the candy cane.
The collisions between water molecules and sugar molecules are crucial for the dissolving process. With higher temperatures, there are more collisions, so more opportunities for sugar molecules to break away from the candy cane and become surrounded by water molecules.
The increased energy and movement at higher temperatures also weakens the intermolecular forces holding the sugar molecules together in the candy cane. As a result, it is easier for water molecules to break these forces and incorporate the sugar into the water, leading to a faster dissolving time.
Try These Candy Cane Activities
- Crystal Candy Canes
- Fluffy Candy Cane Slime
- Candy Candy Slime
- Peppermint Oobleck
- Candy Cane Bath Bombs
- Bending Candy Cane Experiment
- Paper Lollipop



More Fun Dissolving Experiments
- What Dissolves in Water
- Dissolving Gingerbread Cookies
- Dissolving Candy Hearts
- Classic Skittles Science
- Floating M&Ms
Printable Candy Cane STEAM Project Pack
For a complete pack of instructions, templates, and extras, grab our Candy Cane Project Pack!











One Comment
Comments are closed.