Slime is one of the most popular hands-on science activities for kids — but it also raises a lot of questions.
Is slime a liquid or a solid?
Why does it stretch sometimes and snap other times?
What actually happens when you mix glue and activator?
The answers come from basic chemistry concepts kids can actually see and feel. This guide explains what slime is made of and why it behaves the way it does in simple, kid-friendly terms.

Quick Answer: Is Slime a Solid or Liquid?
Slime is neither a true solid nor a true liquid.
- Pull slowly → it flows like a liquid
- Pull quickly → it snaps like a solid
Because it changes behavior under shear, slime is classified as a non-Newtonian fluid.
What Is Slime Made Of?
Slime forms when glue reacts with an activator.
Glue (PVA polymer)
+
Borate ions (activator)
= Slime
Common activators include:
- saline solution
- liquid starch
- borax powder
When combined, the molecules connect in a process called cross-linking.
👉 See how slime activators work: Slime Activator Guide

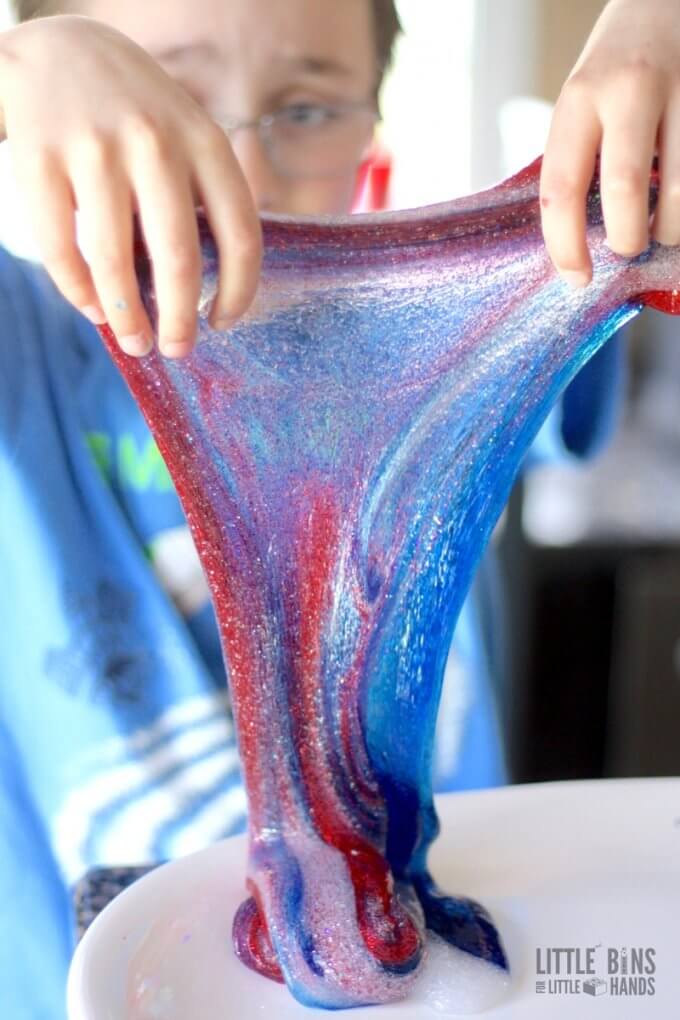
Why Slime Is Stretchy
Glue consists of long, flexible molecular chains that typically slide past one another easily.
When the activator is added, the molecules bind and tangle. The material stops flowing freely and becomes stretchy.
Think of it like:
Fresh-cooked spaghetti → slippery
Cold spaghetti → clumped together
Slime behaves like clumped spaghetti.
Why Slime Breaks (Shear Thickening)
Slime reacts differently depending on how fast you move it.
Pull slowly → stretches
Pull quickly → snaps
Fast movement breaks the molecular bonds.
This property is called shear thickening.
It’s why slime can feel rubbery one moment and gooey the next.

Slime Is a Polymer
A polymer is a long chain of repeating molecules.
Everyday polymers include:
- rubber bands
- plastic containers
- hair gel
Slime is special because children can observe a polymer reaction in real time.
Slime as a Science Activity
Slime demonstrates real science concepts:
- states of matter
- viscosity
- elasticity
- chemical reactions
- material properties
Once kids understand slime changes depending on how they handle it, they naturally want to test it themselves.
👉 Try making slime using our basic recipes: Homemade Slime Recipes
Can You Make Slime Without Borax?
Yes — but it behaves differently.
Taste-safe slime uses thickening agents instead of polymer cross-linking, so it won’t stretch as much as traditional slime.
👉 See borax-free slime ideas

Try a Slime Science Experiment
Change one variable at a time and observe what happens:
- amount of activator
- type of glue
- temperature
- mixing speed
Questions kids can test:
Which slime stretches the farthest?
Which breaks the fastest?
Slime Vocabulary for Kids
Polymer — long-chain molecule
Cross-linking — molecules connecting
Viscosity — how easily something flows
Non-Newtonian fluid — acts like a liquid and a solid
Helpful Slime Resources

Frequently Asked Questions
Why does slime feel rubbery?
The molecules in slime are tightly connected. When you pull it quickly, they resist movement and the slime feels firm or rubbery.
Why does slime melt over time?
The connections between molecules slowly loosen when slime sits without being handled. Kneading the slime again usually restores the texture.
Why is slime sticky?
Sticky slime means not enough activator formed enough cross-links between the glue molecules. Adding small amounts of activator and kneading fixes it.
Is slime safe for kids?
Slime is a chemistry activity and should be used with supervision and proper ingredients. Always wash hands after playing.
Is slime a liquid or a solid?
Slime is neither. It is a non-Newtonian fluid that behaves like a liquid when moved slowly and a solid when force is applied quickly.
Can you make slime without borax?
Yes, but it behaves differently. Borax-free slime uses thickening ingredients instead of a polymer reaction, so it won’t stretch the same way.
The Science Behind Slime (For Curious Kids)
Glue is made of long chains of molecules called polymers. In liquid glue, those chains slide past each other easily, which is why glue pours and spreads.
When an activator containing borate ions is added, the polymer chains begin connecting to each other. This process is called cross-linking. Instead of moving independently, the molecules form a loose network that traps water between them.
Because the water is trapped but not completely locked in place, slime behaves in a unique way:
- move it slowly → the chains slip past each other and it flows
- pull it quickly → the network tightens and resists motion
That’s why slime can drip like a liquid but bounce or snap like a solid.
This special behavior is what scientists call a non-Newtonian fluid — a material that changes how it moves depending on the force applied.
Take Slime Further
Measure and compare:
- stretch distance
- breaking point
- reaction time
- texture changes
Printable Slime Science Guide
Once kids understand why slime stretches, snaps, and flows, the next step is testing it themselves.
This printable Slime Science Guide turns slime into a simple investigation instead of just a one-time activity. Kids compare textures, record results, and experiment with different ingredients while practicing observation skills.
The activities connect to real science concepts like polymers, viscosity, and material properties but stay easy to run at home or in a classroom.
Included:
- investigation pages
- observation charts
- variable testing ideas
- simple explanations kids can understand









For your information, Slime is created when PVA, in other words Polyvinyl acetate, comes in contact with sodium borate, sodium tetraborate, or disodium tetraborate. Slime is a very easy craft that me and my class love so much, that we make it every Monday! We recommend the following slimes: Floam slime, Fluffy slime, Iceburg slime, Crystal clear slime, and Bubble Rap slime.
food color do you use
We use the box of McCormick liquid food coloring bottles from the spices section of the grocery store!
Can you give me some of your slime recipes?
Here’s our best collection of classic recipes.
https://littlebinsforlittlehands.com/how-to-make-slime-recipes-elmers-glue/
HI,
I’m concerned that borax is not good for you. Does the borax react with the PVA to make it less dangerous?
Thanks,
Lisa.
Have you checked out Steve Spangler? He has a great video or two on Facebook regarding the safety of borax powder for slime making.
Why when you add too much borax, the slime becomes stiff? Can u reply ASAP! thank you! 🙂
Borax slime isn’t my favorite. It is a bit more finicky to make and can get crumbly.
there are two different types of borax. one is detergent, and one is to kill insects. the detergent is safe, but NOT the killer one
Actually boric acid which is in the same boron family as borax powder is what is used in insecticides as well as eye drops. Borax powder is laundry, some hand soaps and trace in fertilizer since it is mined through the ground. Through an intense processing borax powder can be turned into boric acid. They are however, not exactly the same.
tried different versions of your recipes, with the saline solution we had at home, and the result is too liquid every time… do all saline solutions contain sodium borate? the label on my saline solution does not mention it, therefore I wonder if that isn’t the reason why it doesn’t work. is there something else I can use instead? i was hoping to avoid borax
Thx
Yes, your saline needs the ingredients boric acid and sodium borate to work properly. Depending on your location, you can also try liquid starch.
slime is amazing you can do anything with it like: Put it in stress balls, or play with it with your hands. Also, it’s a great stress reliever I do recommend this. Thx for the recipes 😀
I need some slime facts, I am doing a project on slime at school. I need some help. Thanks, Everest
Hi there- I want to use your liquid starch slime recipe for my first grade Halloween party–Can you tell me if one recipe is for one kid or for how many—so for a class of 24- do I make 24 recipes?
THANKS
Hi! One recipe will make quite a bit of slime. One batch could easily be split 3-5 ways depending on how much you want each kid to end up having. I like the condiment size containers that hold about 2oz each! One batch usually fills about 5 of those! Feel free to email me sarah@littlebinsforlittlehands.com if you would like to talk about it more!
im using this for my project
As someone who’s read about and experienced PVA and borax together I would say that you should probably stay away from it. I know that there were many reports on the news about kids getting burns from the borax in their slime. Personally I did not get burned but my hands did feel tingly and slightly hot. All in all it does work as an activator but I would advise to choose another activator like detergent or contact lenses solution.
Sensitivity is a huge issue. However, my skin did not like laundry detergent at all and I highly advise to stay away from laundry detergent which is why we do not have a recipe for it. We use less borax than most recipes, and if you do have sensitive skin I would choose saline solution (not contact lens solution). Liquid starch is another choice. All the choices contain some form of borons.