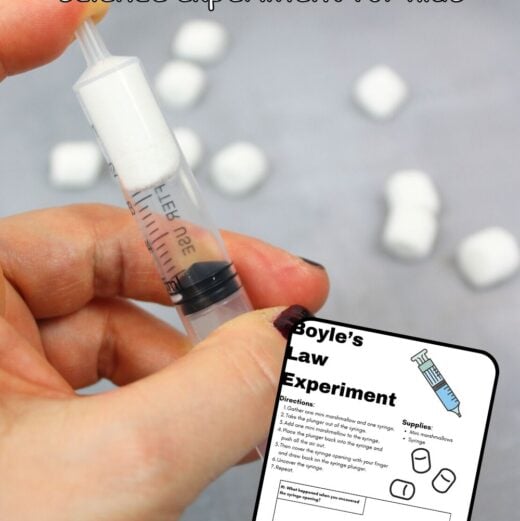
Explore how pressure changes affect volume with this simple Boyle’s Law experiment using mini marshmallows and a syringe! This hands-on science activity lets kids see the invisible science of air pressure in action — no fancy lab required.

What Is Boyle’s Law? (Simple Definition for Kids)
- Field of Science: Physics
- Grade Level: 4th–7th Grade
- Concepts Explored: Air pressure, gas laws, force, and volume
Boyle’s Law explains the relationship between pressure and volume in gases. When the pressure on a gas increases, its volume decreases. When the pressure decreases, its volume increases.
This marshmallow syringe experiment makes it easy to see Boyle’s Law in action — perfect for introducing kids to the gas laws in physics.
Boyle’s Law Air Pressure Experiment Supplies
You Will Need:
- Mini marshmallows
- 1 syringe (without a needle)
This simple setup demonstrates how air pressure and volume are interconnected — all using materials you likely already have at home or in the classroom.
How to Do the Boyle’s Law Experiment Step-by-Step
🎥 Watch the video:
- Gather one mini marshmallow and one syringe.
- Remove the plunger from the syringe.
- Add one mini marshmallow inside.
- Replace the plunger and push most of the air out.
- Cover the syringe opening tightly with your finger.
- Pull back on the plunger and observe what happens.
- Uncover the opening and note what changes.
- Repeat the process to confirm your results.
🔎 This hands-on demonstration helps kids observe how changing pressure changes volume — a key physics concept.


Boyle’s Law Explained with Marshmallows
How does it work? When you pull the plunger back, the pressure inside the syringe decreases, and the air trapped in the marshmallow expands — making it puff up.
When you push the plunger in, pressure increases, and the marshmallow shrinks as the air pockets are compressed.
This is a clear example of Boyle’s Law — the inverse relationship between pressure and volume.
Pressure × Volume = Constant
or
P₁V₁ = P₂V₂
Real-Life Examples of Boyle’s Law
Robert Boyle, an Irish scientist, first described this relationship in the 1600s. His findings still help us understand how gases behave today.
You can find Boyle’s Law in action all around you:
- In weather: Changing air pressure creates winds and storm systems.
- In scuba diving: Divers experience pressure changes as they move deeper underwater.
- In medicine: Syringes and lungs both rely on air pressure differences.
🔎 This Boyle’s Law demonstration makes these invisible forces visible for kids. DIY Scientific models are a fantastic way to illustrate big-picture concepts like this one.
Hands-On Physics Activity: Make It a Science Project
Turn your marshmallow experiment into a Boyle’s Law science project!
Try testing these variables:
- Use different sizes or brands of marshmallows.
- Try other compressible materials like foam or gummy candy.
- Vary how much air you leave inside the syringe.
Ask questions like:
- How does plunger movement affect marshmallow size?
- Does temperature change the results?
- What would happen if you use a smaller or larger syringe?
🔎 Add the scientific method (explained here)! Encourage students to make predictions, collect observations, and record results using a data sheet or printable worksheet.
NGSS Guiding Questions (Grades 4–7)
| NGSS Question | Concept Focus |
|---|---|
| What happens to matter when conditions like pressure change? | Matter changes shape and volume when external pressure changes. |
| How can we use patterns to explain natural phenomena? | The relationship between pressure and volume can predict how gases behave. |
| How do scientists use models to understand invisible forces? | A syringe acts as a model to visualize how gas molecules behave under different pressures. |
Related Science Experiments
- Cartesian Diver Experiment
- DIY Barometer Project
- Homemade Lung Model
- Water Bottle Rocket STEM Challenge
- Crushed Can Experiment
- Cold and Hot Air Balloon Experiment



Printable Science Packs for Kids
Visit our SHOP for easy-to-print STEM and science lesson plans for the whole year. Or join us in the Little Bins Club.